Toward to Logical and Integrative Understaing of Human
Department of Integrative and Systems Physiology
Research
Outline
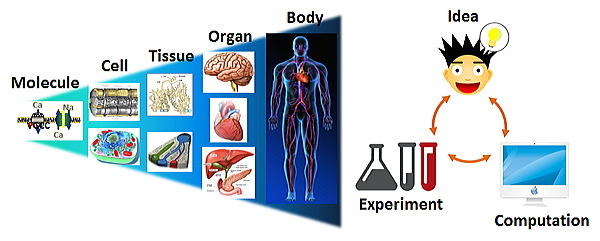
Integrative and Systems Physiology
We have been studying physiology at molecular, cellular and system levels, utilizing techniques combining experimental and mathematical methods, to clarify mechanisms of life and to seek novel strategies controlling living body.
Major projects
1. Study on the mechanisms underlying cardiac physiology/pathophysiology, focusing on intracellular cellular ion dynamics
Intracellular ion concentrations dynamically change (cellular ion dynamics) and are pivotal factors controlling excitation-contraction coupling of ordinary cardiac muscle cells (ventricular and atrial cells). They also play important roles in making a rhythm of specialized cardiac muscle cells (sinoatrial node and atrioventricular node cells). We have been studying the mechanisms of the cellular ion dynamics and seeking new strategies for controlling cardiac cell functions.
a) Experiments on cell physiology
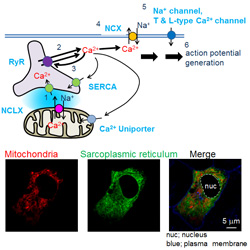
We study how individual molecules, such as channels/transporters, control cardiac cell functions using freshly isolated ventricular, atrial and sinoatrial node cells as well as cardiac cell
line HL-1. We perform electrophysiological experiments, cell motion analyses as well as imaging analyses using fluorescence dyes or proteins in the normal cells and the specific gene
knockout/knockdown cells.
b) Systems physiological analysis using mathematical models
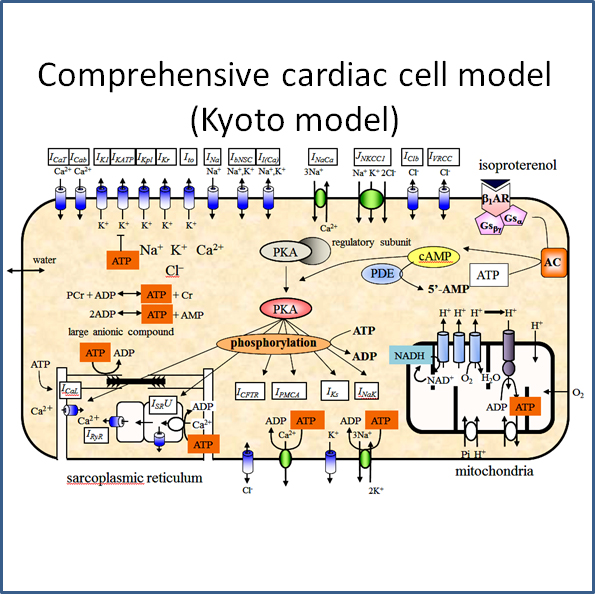
To overcome the limitation of experimental approach and to integrate a large body of scientific findings, we develop comprehensive mathematical models of cells and tissues based on the
experimental data, and perform simulation analysis so as to clarify the underlying mechanisms. The representative models are models of ventricular cell, atrial cell, sinoatrial nodal
cell and HL-1 cell. By repeating a cycle of “development of simulation-based working hypothesis” and “experimental validation”, we have clarified quantitatively contribution of
each component of the cell on the cardiac cellular functions. In addition, we analyze how an abnormal activity of a component causes the failure of cellular function.
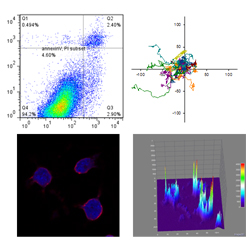
2. Study on the mechanisms of ion dynamics of lymphocytes and their role in pathophysiology
Cytoplasmic Ca of lymphocytes increases upon various stimulations such
as antigen binding to antigen receptors. We study the mechanisms of ion
dynamics of lymphocytes and the relationship between ion dynamics and immune
abnormality. We succeeded in developing a comprehensive model of Ca dynamics
of lymphocyte and elucidated that a novel mitochondrial Ca transporter
plays a major role in immunological responses of B lymphocytes.
3. Study on the dynamic mechanisms of gating in ion channel proteins.
Ion channels are regulated by various kinds of chemical and physical stimuli such as ligands, voltages, and etc. We study the dynamic mechanism underlie the activation by the diffracted x-ray tracking, which can trace conformational changes of the channels at single-molecule level. We previously reported twisting motions of KcsA potassium channels uopn gating at video rate. By combining the development of the recording methods and the data analysis we are trying to reveal the common mechanism for the activations at further high-temporal and spatial resolutions.